The Unified eClinical Platform that ambitious clinical teams switch to.
All-in-one eClinical platform and data management solutions for modern clinical trials.
Intuitive, flexible, and built to streamline study setup, data collection, and oversight without the complexity of legacy systems.
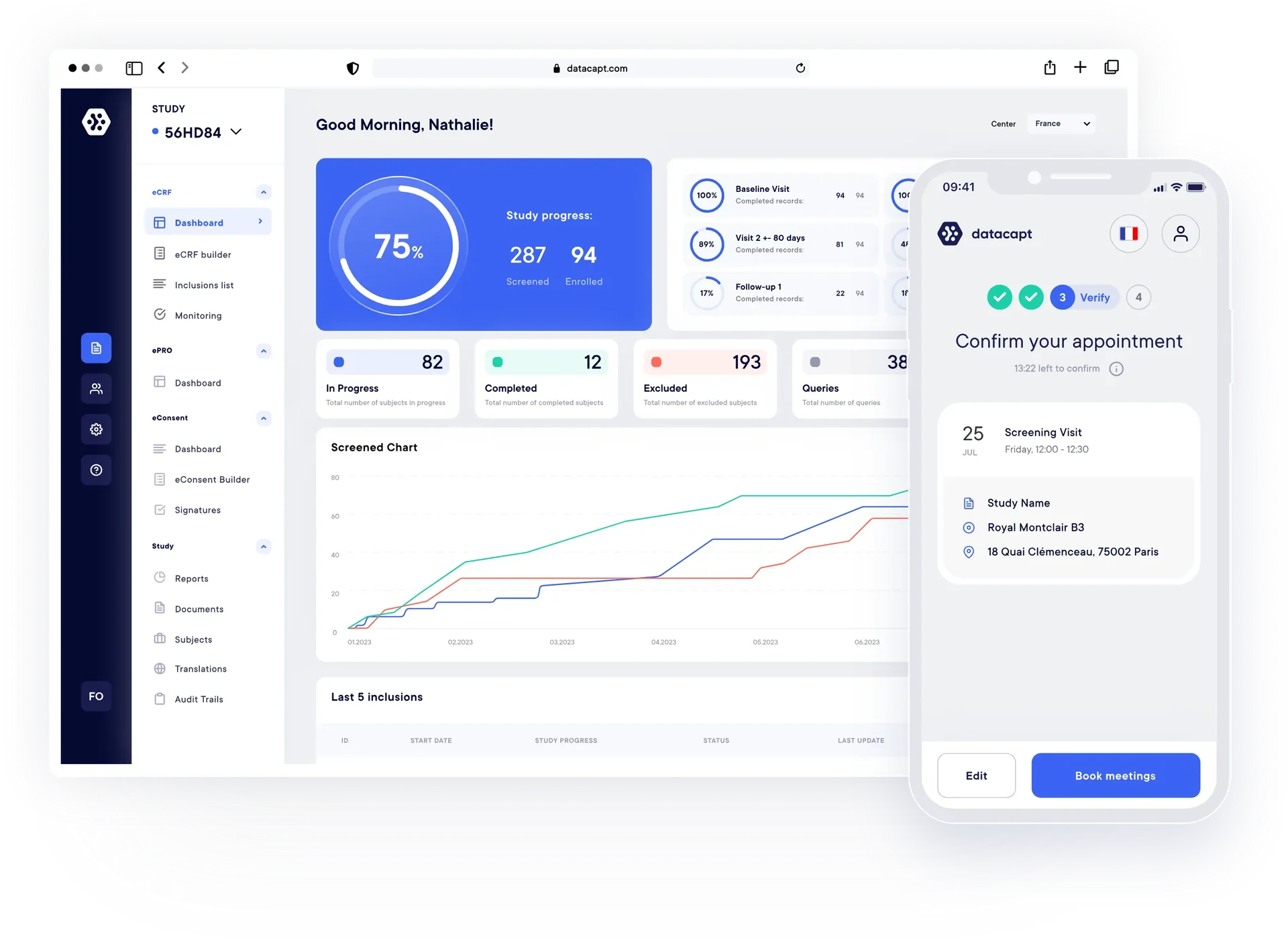
Our Solutions.
EDC / eCRF
Build studies in days, no coding required. Collect and monitor everything in one place for clean, reliable data.

ePRO Surveys
Patient-reported outcomes, captured in real-time. Send surveys to any phone, automated reminders, no transcription.

eConsent Signature
Better comprehension, better retention. Patients review the consent at their own pace and sign from anywhere.

Randomization
Stratified, block, or minimization. Fully configurable, randomization linked directly to the eCRF/EDC.

Medical Coding
Code adverse events and medications in one workflow. Ranked suggestions per verbatim term, approved in one click.

Trial Supply Management
Every site has the right kit, every visit. Per-site forecasting, automatic resupply triggers, and dispensing linked to the eCRF.

Integrations API
Talks to every system you already run. REST APIs, webhooks, real-time exports, SSO, labs, safety, CTMS, and warehouse, all connected.

Televisits
Run the visit, capture the data, all in one place. Compliant video inside the eCRF, eSource captured live, fewer no-show deviations.

Participant Database
Easily register, manage, and track research participants. Streamline recruitment, consent, and study coordination from a single dashboard.

Scheduling
Automate visit scheduling, calendar sync, and reminders for efficient site operations.

Recruitment
Accelerate patient enrollment with advanced pre-screening and real-time recruitment tools.

Payments
Simplify patient reimbursements and track payments seamlessly within CTMS.

Trusted. Smart. Ready.
Datacapt revolutionizes clinical trial management, making it accessible to all industries
and enabling teams to deliver results with unmatched speed and efficiency.
Studies and +6,000 Sites
Faster (design, collect, reports)
Countries and +85 Languages
Designed for Every Study,
Built for your Needs.
Datacapt
for CROs
They already use datacapt





Datacapt
for Medical Devices
They already use datacapt





Datacapt
for Biotech and Pharma
They already use datacapt




Datacapt
for Academic Research
They already use datacapt





Datacapt
for Consumer Health
They already use datacapt
.webp)
.webp)




Datacapt surpassed our expectations thanks to its user-friendly interface and the availability of its team. It made form design and data collection remarkably intuitive resulting in significant time savings and enhanced accuracy.

Want to see Datacapt in action?
Dive into our interactive demo and explore how Datacapt simplifies every step of your clinical study from eCRF to scheduling, eConsent, reporting, and more. No jargon, no commitment, just a hands-on tour of the platform in action.
Why choose Datacapt?
Scalable and compliant
Stay on top of security
.webp)

3 billion +
Data points efficiently and securely managed


99.99 %
Uptime

50 000 +
End users globally

What do customers say about Datacapt?






.webp)















.webp)









.webp)

Built for Trials.
Powered by Trust.
Experience the Difference.
Blog & News Datacapt
News, Articles, Resources and Tutorials.












.webp)
.webp)
